
About this pulmonary arterial
hypertension clinical trial
••••••
What happens in pulmonary arterial hypertension?
In pulmonary arterial hypertension, or PAH for short, the pressure in the blood vessels in the lungs is too high. As a result, people with PAH can experience breathlessness and tiredness during increased physical activity.
What is this clinical trial in pulmonary arterial hypertension looking at?
This PAH clinical trial is called VIPAH•PRN2B
The VIPAH•PRN clinical trial2B is looking at an investigational treatment called RT234 for people with PAH.
This Phase 2, open-label trial will:
Continue to monitor the safety of RT234
Test whether RT234 can help to reduce breathlessness and tiredness during increased physical activity in people with PAH
Test what the best dose of RT234 is
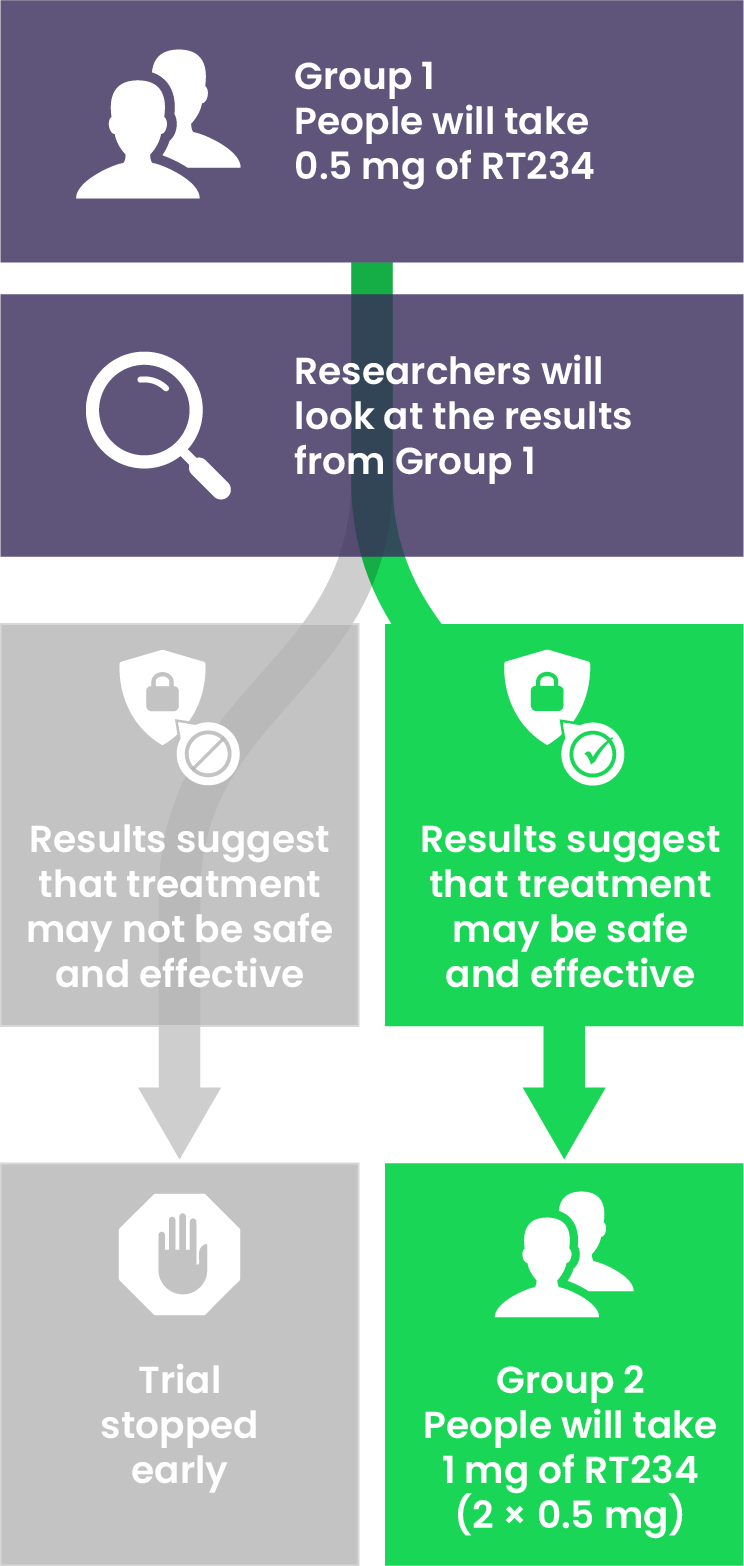
What will happen during the VIPAH•PRN2B trial?
There are 2 treatment groups:
Each person will only participate once, either as part of Group 1 or Group 2.

People in each group will be asked to:
Participate in:
visits in
the clinic
appointment
over the phone
Complete their involvement in:
days
Participants will be asked to perform different activities to allow researchers to compare how well they perform during exercise tests without RT234 and after taking it. The VIPAH•PRN2B trial will also look at the side effects of RT234.
Activities during the trial:
6-minute walk test (×3)
Safety assessments (×3)
RT234 inhalation (×2)
Multiple blood samples
will be taken during 3 visits
Cardiopulmonary exercise testing (×2)
To see the schedule of activities
What is RT234?
••••••
RT234 is an inhaler and a capsule. The capsule is filled with medicine in a form of a dry powder.
This capsule is placed inside the inhaler before use. The inhaler was specifically designed to deliver the powdered medicine to your lungs as you breathe it in.
RT234 is tested as a treatment that people can inhale when they need it, for example, before they exercise or perform different daily activities.

RT234 is still being studied and has not yet been approved for doctors to prescribe to people with PAH.
How do I know if I can take part?
••••••
You may be able to take part in this trial if:
You are 18-80 years of age
You have WHO Group 1 PAH, which was diagnosed using a test called right heart catheterization
Your doctor ruled out the possibility that you have PAH caused by long-term blood clots in the lungs (known as chronic thromboembolic pulmonary hypertension)
You have not been abusing alcohol, drugs, or solvents in the last 6 months
Your doctor determines that you can take part based on how well your lungs work during cardiopulmonary exercise testing at screening
You are on stable treatments of up to 3 medications for PAH, that you take by mouth or via inhalation. You cannot receive any injected medications for PAH (including intravenous or subcutaneous medications), or certain tablets for PAH
About Respira Therapeutics
••••••
Respira is a company developing next-generation inhaled therapies that target the lungs. Our aim is to address the significant unmet needs of people with cardiopulmonary diseases. We have developed an investigational dry-powder inhalation device and applied advanced particle engineering technologies to create a drug-device product that is being studied for potential use by people with PAH.
Patients are at the forefront of everything we do, and we are committed to bringing innovation to people living with PAH.
To learn more about Respira

